A research team led by Kathy Luo Qian, professor in the Faculty of Health Sciences (FHS) at the University of Macau (UM), has developed new models of muscle atrophy using sensor zebrafish. The study shows that atrophied muscles can improve overall muscle mass and strength by increasing the size of individual muscle cells, ultimately improving patients’ mobility and health. These findings offer new insights and ideas for the treatment of muscle atrophy. The research has been published in the internationally renowned journal Advanced Science.
Muscle atrophy can occur as a result of a number of conditions, including aging, prolonged bed rest, starvation, nerve injury, or diseases such as cancer, leading to reduced mobility. Over the past few decades, many methods have been used to treat muscle atrophy, including exercise, electrical stimulation, ultrasound therapy, and nutritional therapy. However, the results of these methods are not satisfactory. In muscle atrophy, the reduction in muscle mass can be attributed to either the death of muscle cells or the shrinkage of individual muscle fibres. Understanding this is therefore crucial to the treatment of diseases associated with muscle atrophy and to improving health outcomes.
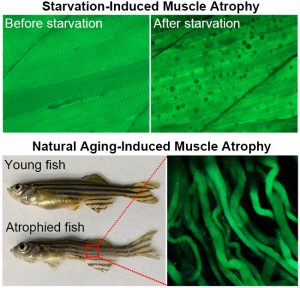
To accurately detect muscle cell apoptosis, the research team developed a method for real-time tracking of muscle cell apoptosis in live zebrafish. They specifically expressed a genetically encoded fluorescent protein sensor in the muscle tissues of zebrafish. The sensor emits green fluorescence in live cells and blue fluorescence in apoptotic cells. Using these sensor zebrafish, the team found that in starvation-induced muscle atrophy, muscle cells shrank significantly but did not die. These atrophied muscle cells survived by engulfing their own cellular components, a process known as autophagy. When the zebrafish were refed, the autophagic vacuoles disappeared, indicating that muscle atrophy is reversible.
Zebrafish typically live for three to five years. To study aging-induced muscle atrophy, the research team followed zebrafish with fluorescent sensors for five years. They found that in aged zebrafish with muscle atrophy, muscle cells remained viable despite a 57% reduction in diameter. Gene expression analysis revealed a significant downregulation of apoptosis-related genes and an upregulation of autophagy-related genes.
The study found that in muscle atrophy caused by starvation and aging, muscle cells do not die but remain alive through autophagy. It also suggests that increasing the size of individual muscle cells can increase overall muscle mass and strength, ultimately improving patients’ mobility and health.
Prof Luo is the corresponding author of this study, and Jia Hao, a postdoctoral researcher in FHS, is the first author. PhD graduates Wu Renfei and Yang Hongmei also contributed to the research. The study received important support from the Animal Research Core and the Biological Imaging and Stem Cell Core of FHS. The research was funded by UM (File No.: MYRG2020-00121-FHS and MYRG2022-00025-FHS), the Science and Technology Development Fund of the Macao SAR (File No.: 0147/2020/A3, 044/2021/APD, and 0004/2021/AKP), and the Ministry of Education Frontiers Science Center for Precision Oncology, UM (File No.: SP2021-00001-FSCPO and SP2023-00001-FSCPO). The full text of the research article is available at https://advanced.onlinelibrary.wiley.com/doi/full/10.1002/advs.202416811.
| Source: Faculty of Health Sciences | |
| Media Contact Information: | |
| Communications Office, University of Macau | |
| Albee Lei | Tel: (853) 8822 8004 |
| Jason Leong | Tel: (853) 8822 8322 |
| Email: | prs.media@um.edu.mo |